SOLVED: A balloon is filled with 3.60 L of H2 gas at STP. If the balloon is taken into a sea where the pressure is 2.50 atm and the temperature is 10.00
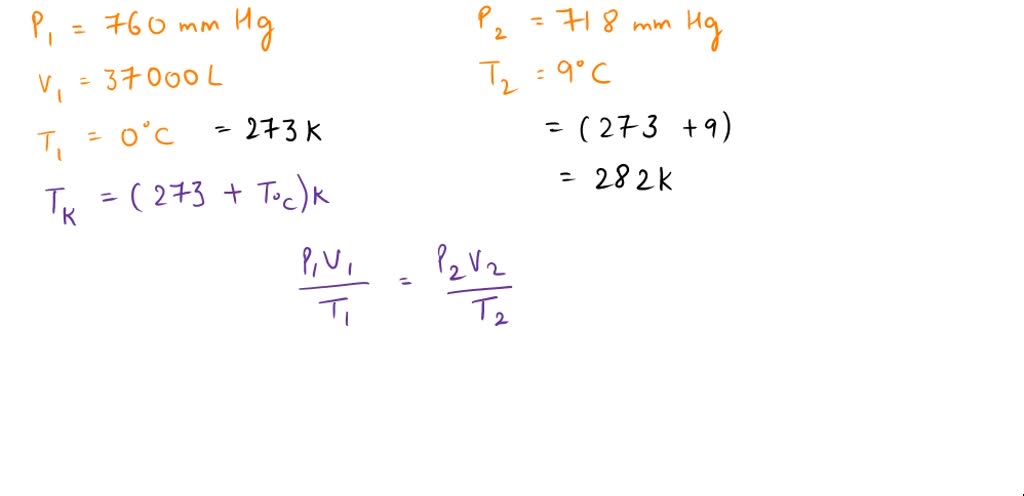
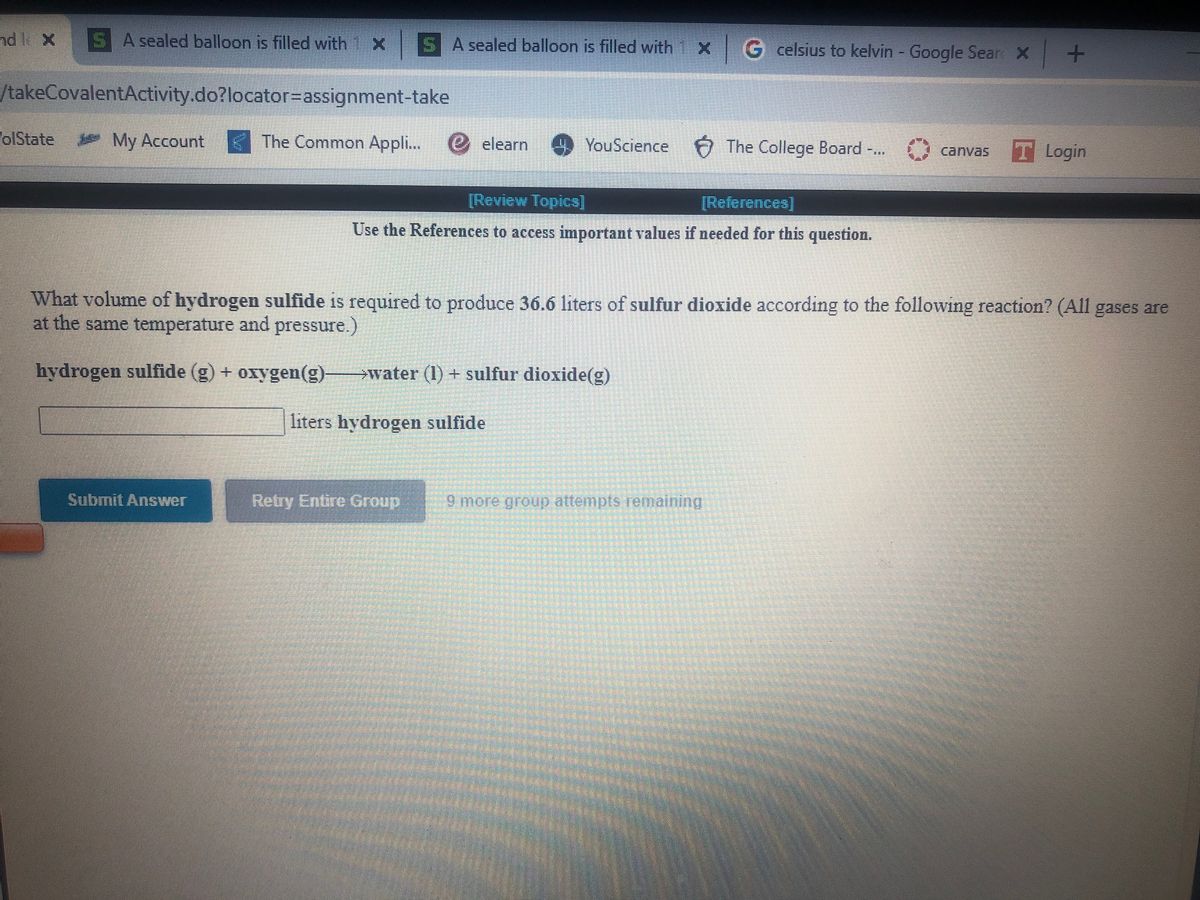
SOLVED: A balloon filled with hydrogen gas had a volume of 37000 L at a pressure of 760 mmHg and a temperature of 0 °C. When the balloon reached an altitude of

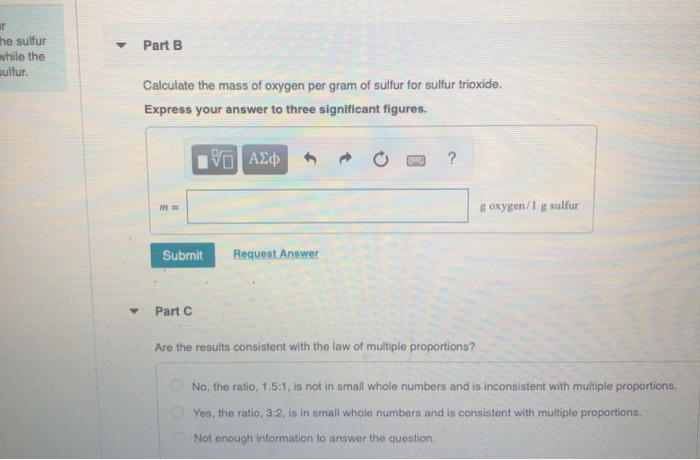
BIOLOGYHonors - Gases Workbook Packet Answers.docx - For All Problems In This Packet Show Your Work. Remember Every Number Needs A Unit And A Substance. Circle Your | Course Hero

9.81 | A balloon filled with helium gas takes 6 hours to deflate to 50% of its original volume. How - YouTube

At a certain temperature, the Kp for the decomposition of H2S is 0.722. H2S(g) arrow H2(g) + S(g) Initially, only H2S is present at a pressure of 0.300 atm in a closed

A balloon partially filled with helium has a volume of `30 m^3`, at the earth\'s surface, where ... - YouTube