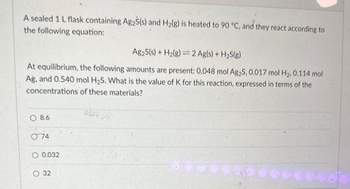
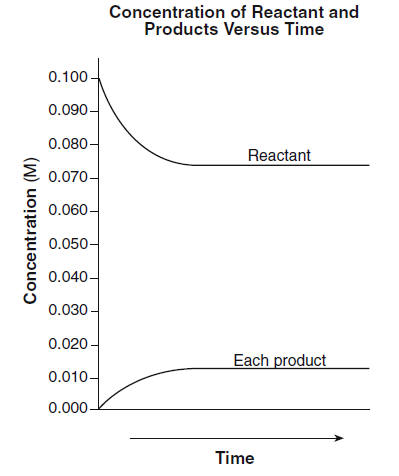
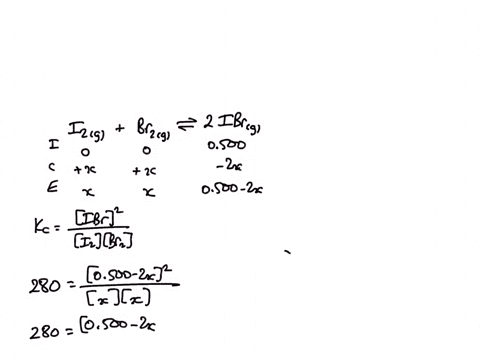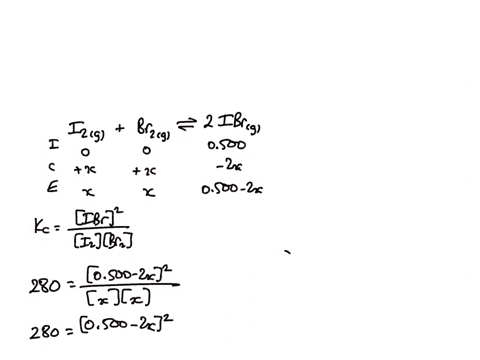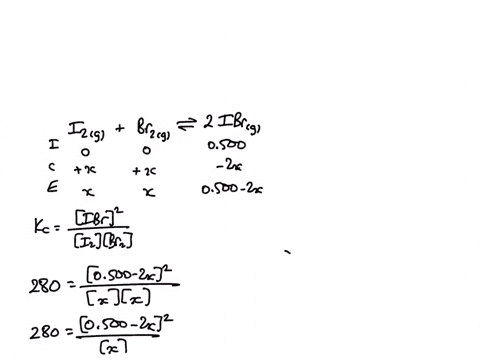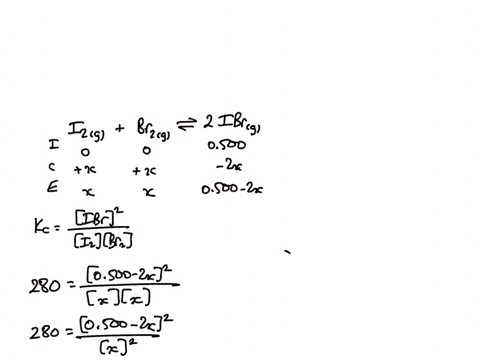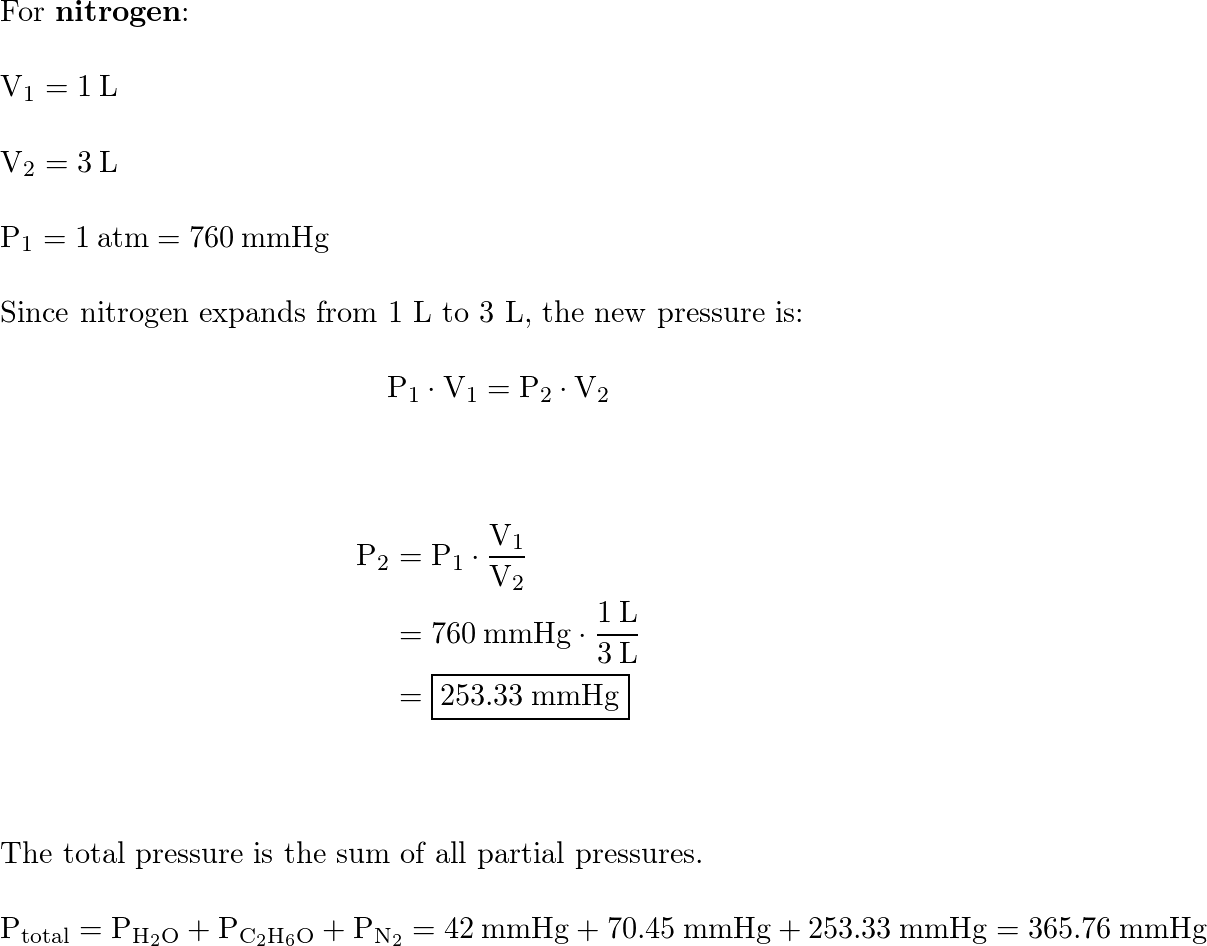1) A sealed 1.0 (L) flask is charged with .500 mol of (I2) and .500 mol of (BR2) an equalibrium reaction ensues: I2 (g) + Br2(G) 2IBr (g) T=120 degree C When

A sealed 10.0 L flask at 400 K contains equimolar amounts of ethane and propanol in gaseous form. Which of - Brainly.com

Synthesis and Reactions of Molybdenum Triamidoamine Complexes Containing Hexaisopropylterphenyl Substituents | Inorganic Chemistry

A sealed 1.0 l flask is charged with 0.500 mol of i2 and 0.500 mol of br2. an equilibrium reaction ensues: - Brainly.com

SOLVED: A sealed 1.0 L flask is charged with 0.500 mol of I2 and 0.500 mol of Br2. An equilibrium reaction ensues: I2 (g) + Br2 (g) ⇌ 2IBr (g) When the

SOLVED: A sealed 1.0 L flask is charged with 0.500 mol of Br2 and 0.500 mol of I2. An equilibrium reaction ensues: I2 (g) + Br2 (g) <—–> 2IBr (g). When the