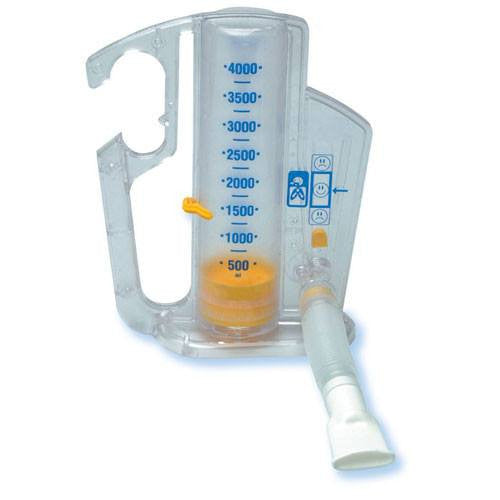
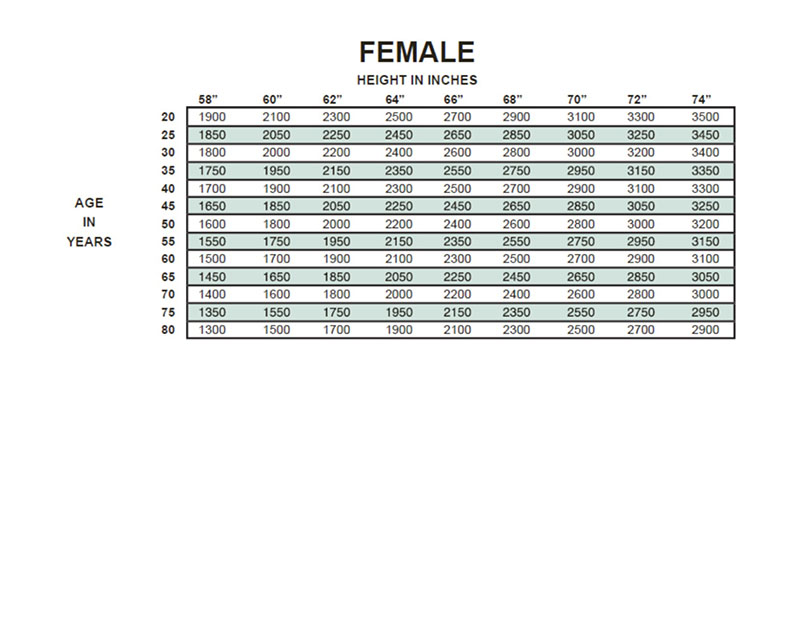
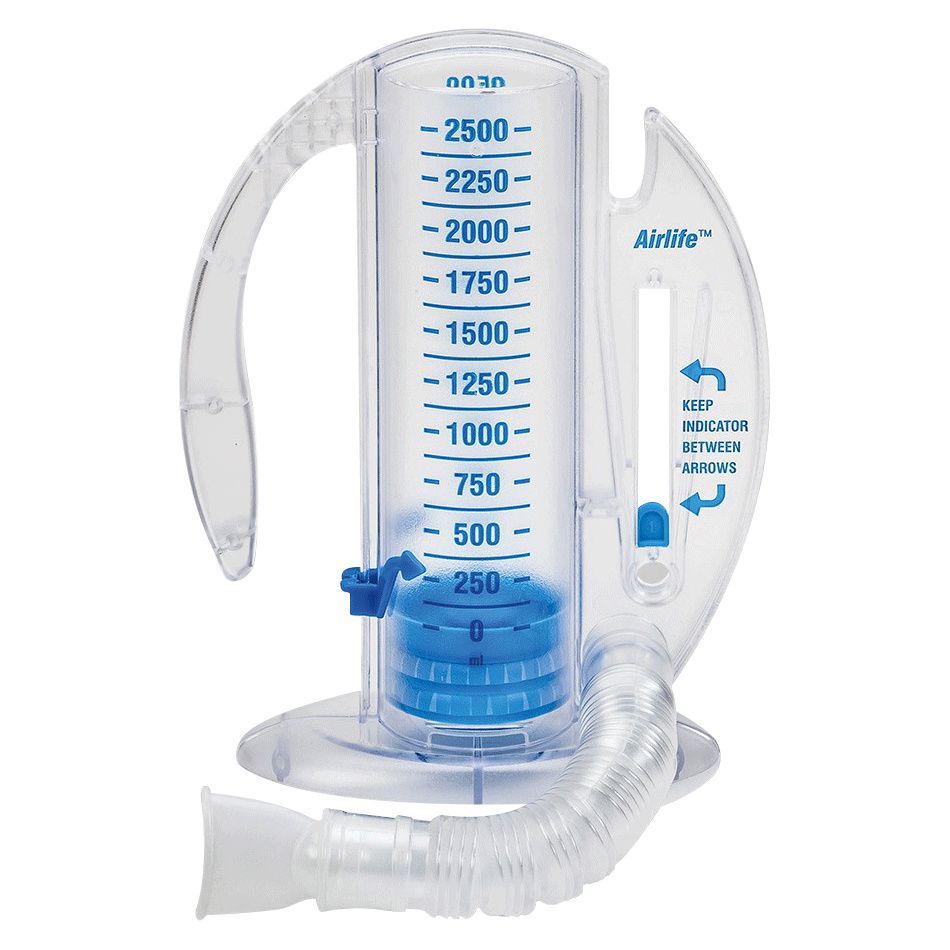
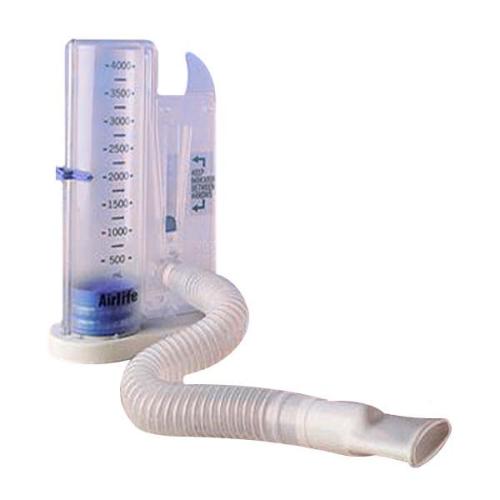
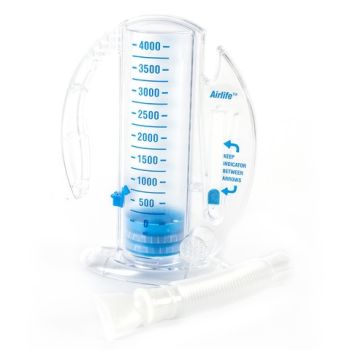
AirLife Volumetric Incentive Spirometer, with 1-way valve (12/cs) - 001902A - Venture Respiratory Inc.
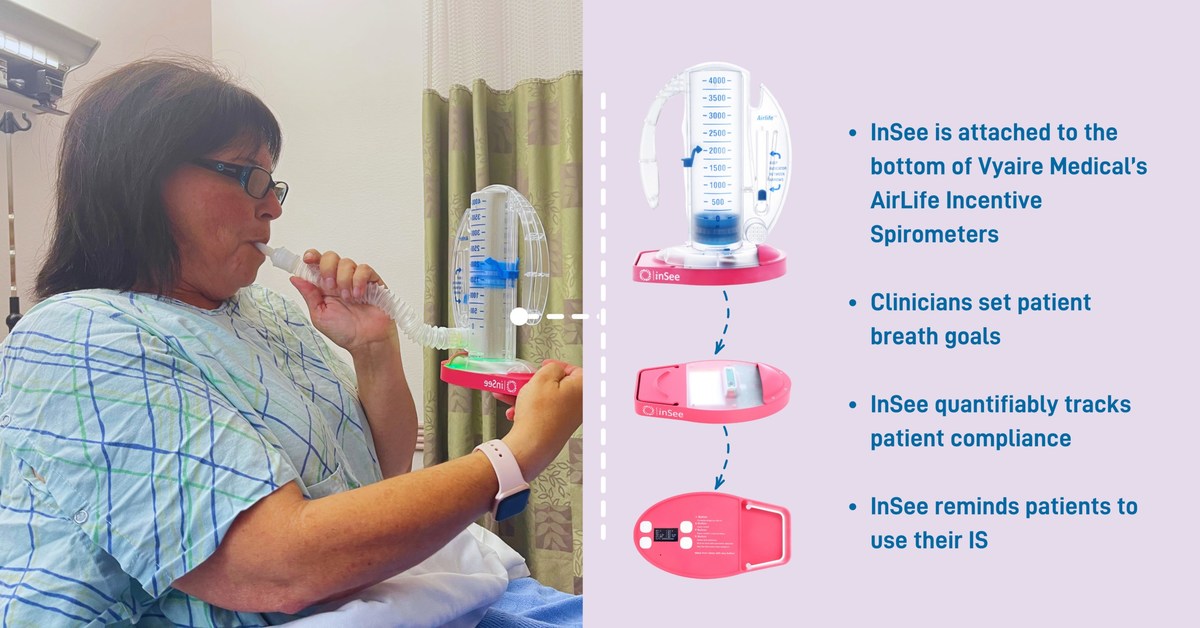
Tidal Medical Technologies receives FDA Emergency Use Authorization (EUA) for the use of InSee for tracking of incentive spirometers usage in treatment of respiratory conditions in COVID-19 patients.

AirLife Volumetric Incentive Spirometer with One-Way Valve, Ball Indicator, 4000 mL, Adult, 1 Count - Walmart.com

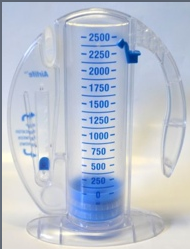
Amazon.com: Vyaire Medical AirLife Volumetric Incentive Spirometer with One-Way Valve, 2500ML, Case of 12, 001903A : Industrial & Scientific
Carefusion Airlife Volumetric Incentive Spirometer Lung Exercise Device ON SALE with Unbeatable Prices




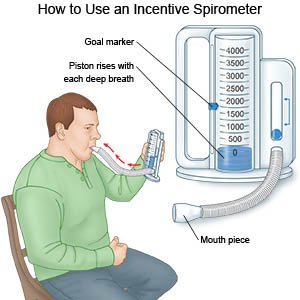





.jpg)