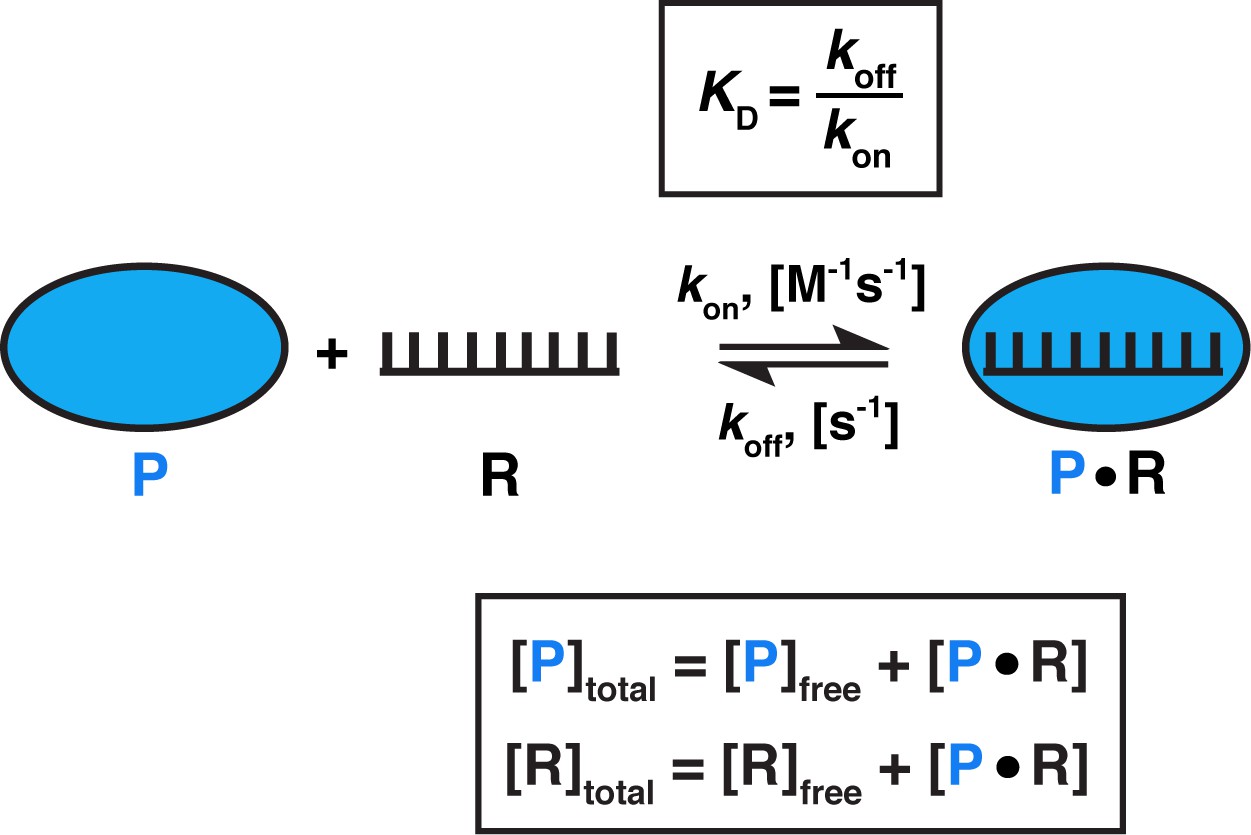
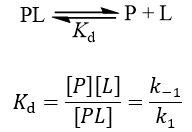![SOLVED: To study binding, we can often determine the fraction of occupied binding sites (Y or 0): binding sites occupied [PL] / total binding sites [PL] + [P]. We can derive a SOLVED: To study binding, we can often determine the fraction of occupied binding sites (Y or 0): binding sites occupied [PL] / total binding sites [PL] + [P]. We can derive a](https://cdn.numerade.com/ask_images/51e8ddf597664cada49ce3b0ea9f0773.jpg)
SOLVED: To study binding, we can often determine the fraction of occupied binding sites (Y or 0): binding sites occupied [PL] / total binding sites [PL] + [P]. We can derive a

Determination of the Binding Constant between Oligonucleotide-Coupled Magnetic Microspheres and Target DNA | ACS Omega

Which software can be used to determine dissociation constant for enzyme-ligand binding using QUADRATIC equation as shown in the pic attached? | ResearchGate

Abbreviations: Ka = affinity (or binding) constant; KD = dissociation... | Download Scientific Diagram

SOLVED: 11 Which one of the following statements about protein-ligand binding is correct? A) The K (association constant) is equal to the concentration of ligand when all of the binding sites are