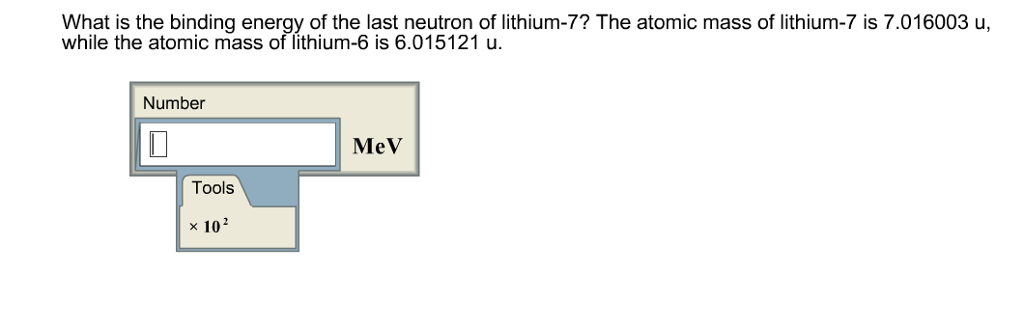
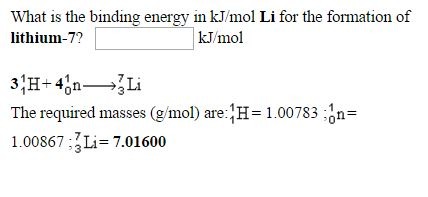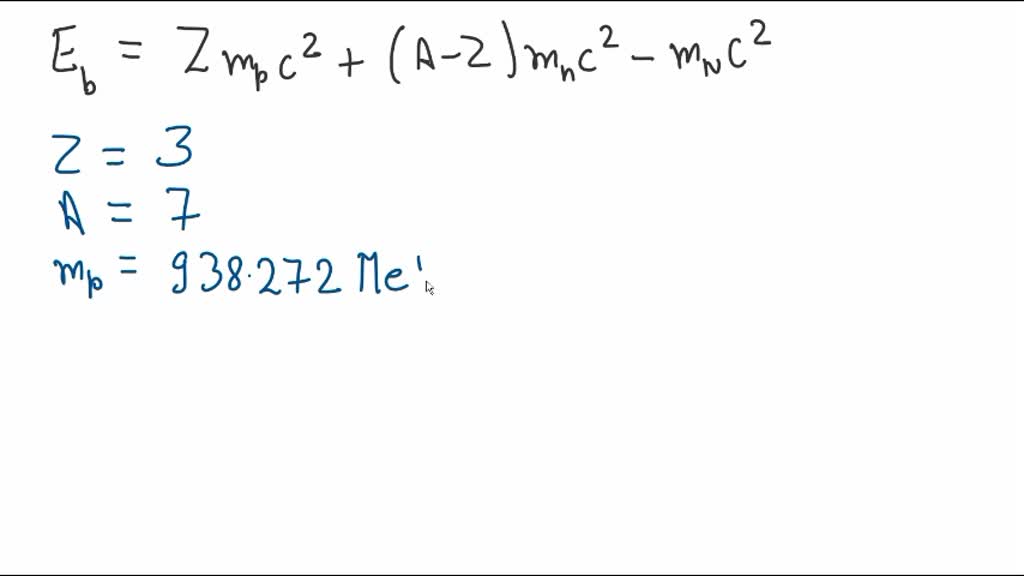The mass of a ^73Li nucleus is 0.042 u less than the sum of the masses of all its nucleons. The binding energy per nucleon of ^73Li nucleus is nearly

Topic – Physics 2a Mass defect and binding energy Prior learning Atomic structure Electrical forces Key words –Atomic nucleus,mass difference, mass of. - ppt download

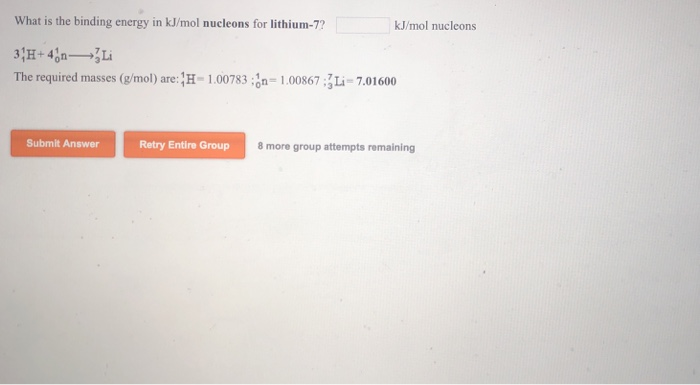
SOLVED: Calculate the nuclear binding energy in joules/nucleon for Lithium-7 (Exact mass 7.01600 amu).

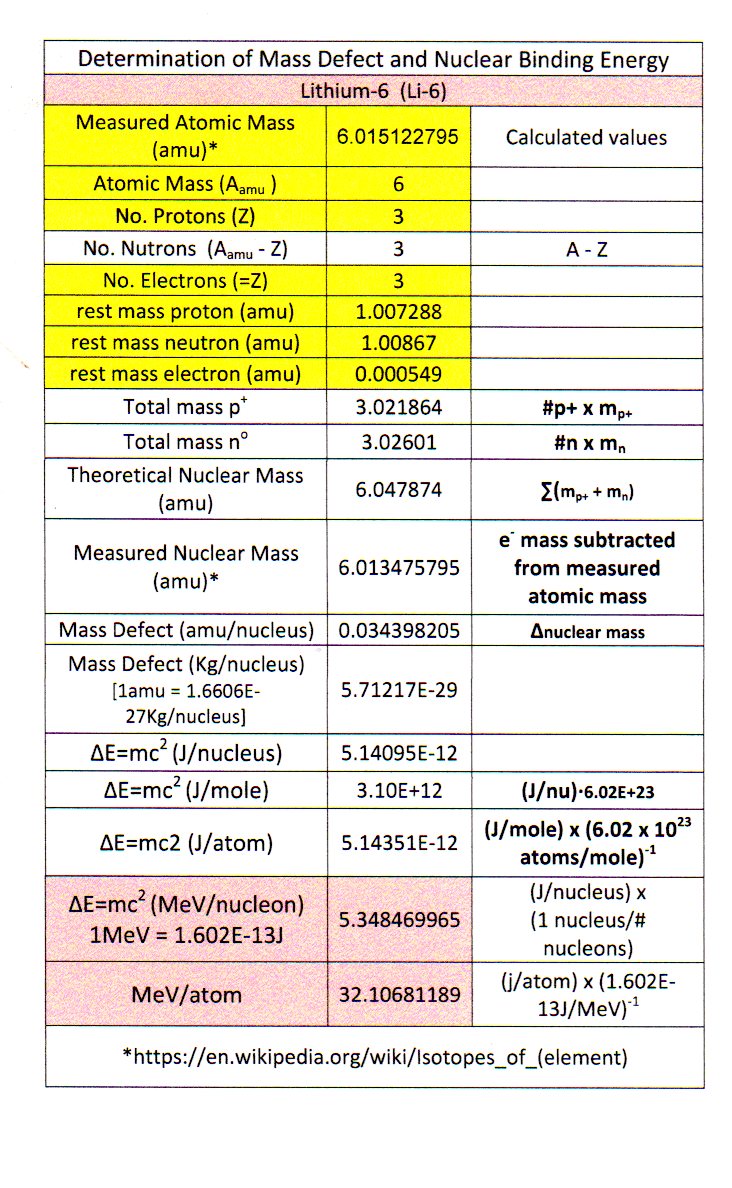
Calculate the (i) mass defect, (ii) binding energy and (iii) the binding energy per nucleon for a 6C^12 nucleus. Nuclear mass of 6C^12 = 12.000000 a.m.u., mass of hydrogen nucleus = 1.007825

SOLVED:What is the nuclear binding energy of a lithium- 7 nucleus in units of kJ/mol and eV/nucleus? (Mass of a lithium- 7 atom = 7.016003 amu.)

SOLVED: Calculate the nuclear binding energy in joules/nucleon for Lithium-7 (Exact mass 7.01600 amu).

If the binding energy per nucleon in `L i^7` and `He^4` nuclei are respectively `5.60 MeV` and ` - YouTube

The energy of the reaction Li ^ 7 + p ⟶ 2He^4 is (the binding energy per nucleon in Li^7 and He^4 nuclei are 5.60 and 7.06 MeV respectively.)